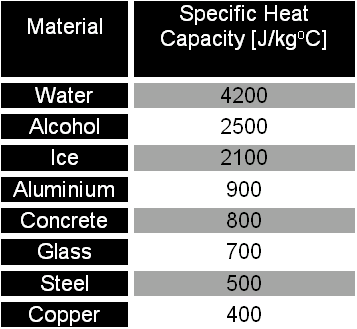
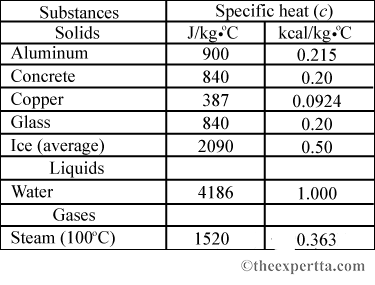
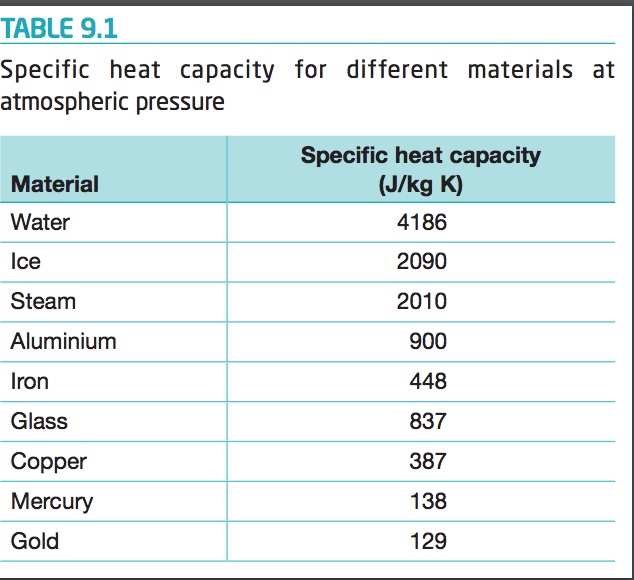
Specific Heat Capacity Measurement for Porous Composite Material with Glass Fiber/Fumed Silica Nanoparticles | Semantic Scholar

Breakdown of One-to-One Correspondence in Energy and Volume in a High-Pressure Heat-Treated Zr-Based Metallic Glass During Annealing | Scientific Reports
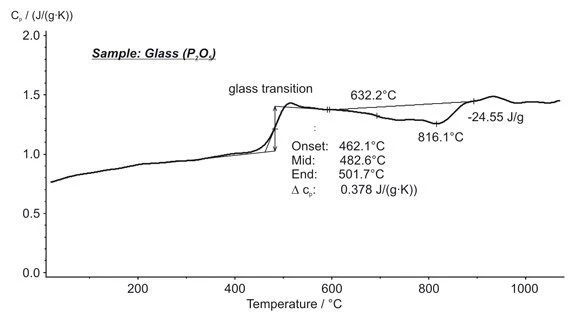
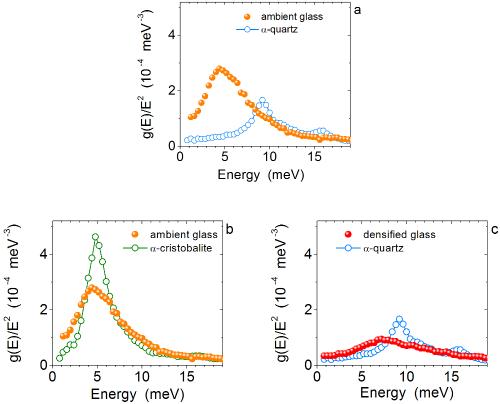
Phosphate Glass Powder — Glass Transition, Structural Change, Specific Heat - NETZSCH Analyzing & Testing
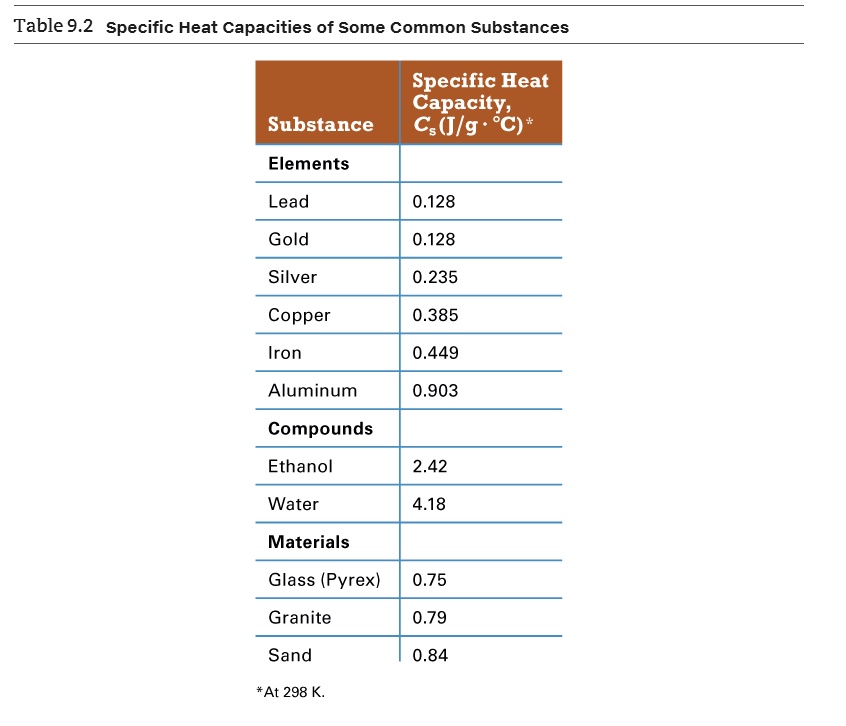
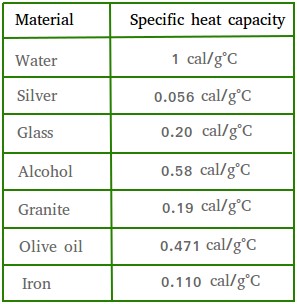
SOLVED: Table 9.2 Specific Heat Capacities of Some Common Substances Specific Heat Capacity; Cs(Jlg. *C) Substance Elements Lead 0.128 Gold 0.128 Silver 0.235 Copper 0.385 Iron 0.449 Aluminum 0.903 Compounds Ethanol 2.42

Specific heat capacities for an as-deposited thin toluene film (solid... | Download Scientific Diagram
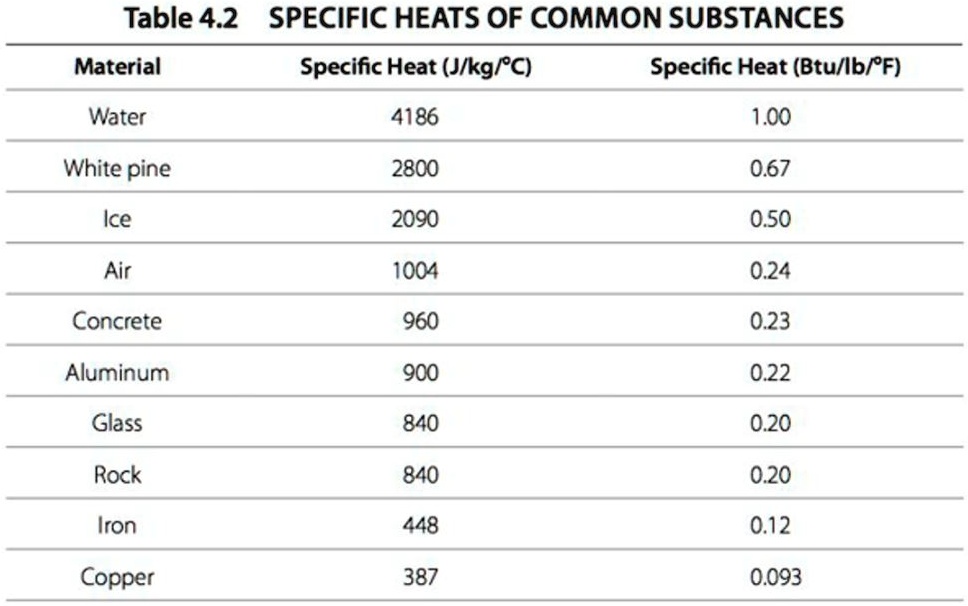
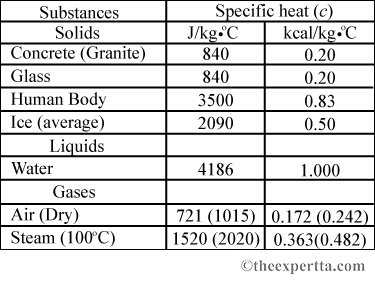
SOLVED: Table 4.2 SPECIFIC HEATS OF COMMON SUBSTANCES Material Specific Heat (JlkgrC) Specific Heat (BtullbrF) Water 4186 1.00 White pine 2800 0.67 Ice 2090 0.50 Air 1004 0.24 Concrete 960 0.23 Aluminum 900 0.22 Glass 840 0.20 Rock 840 0.20 Iron 448 ...

Heat capacity (C p ) vs. temperature curves in the glass transition... | Download Scientific Diagram

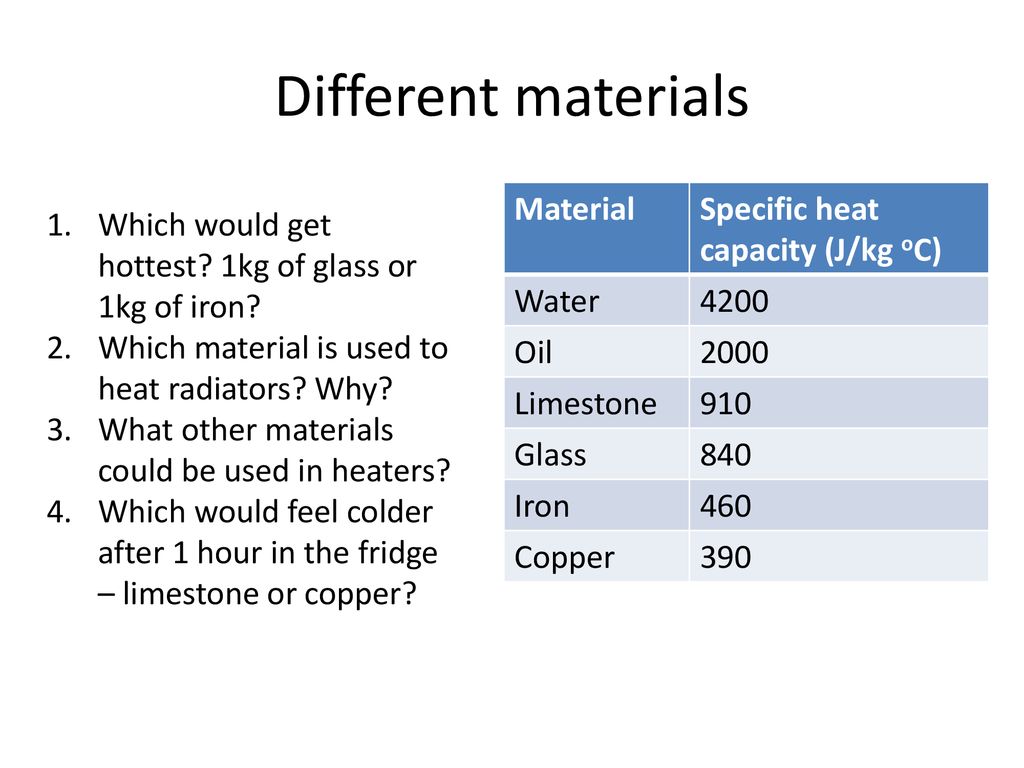
Specific heat capacity Physics Homework Help, Physics Assignments and Projects Help, Assignments Tutors online