Role of the Deposition Precursor Molecules in Defining Oxidation State of Deposited Copper in Surface Reduction Reactions on H-Terminated Si(111) Surface | The Journal of Physical Chemistry C
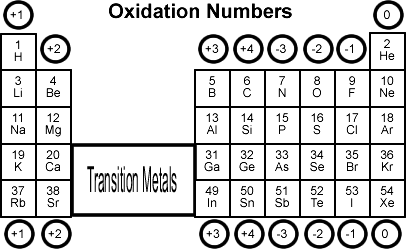
![chemicalperiodicity [licensed for non-commercial use only] / Oxidation number of the oxides and chlorides chemicalperiodicity [licensed for non-commercial use only] / Oxidation number of the oxides and chlorides](http://chemicalperiodicity.pbworks.com/f/1325603788/Summary%20of%20oxidation%20no.%20of%20oxides%20and%20chlorides.jpg)
chemicalperiodicity [licensed for non-commercial use only] / Oxidation number of the oxides and chlorides
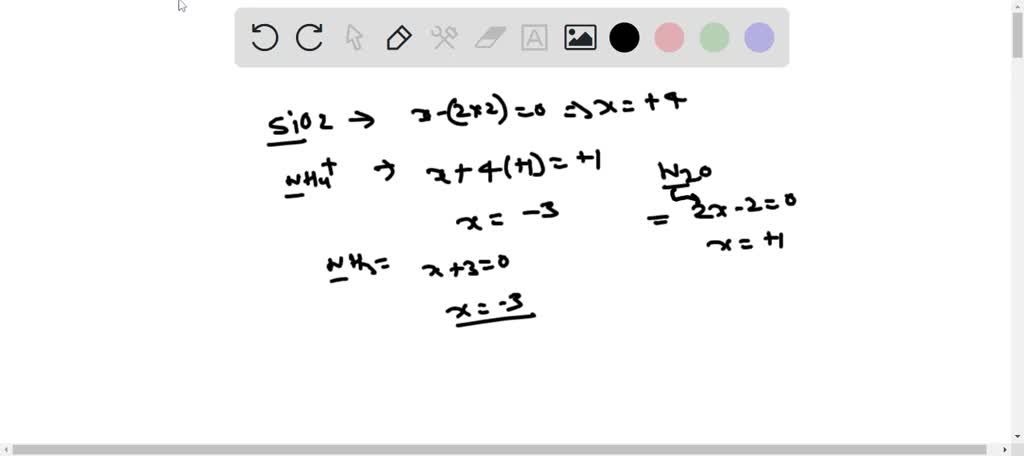
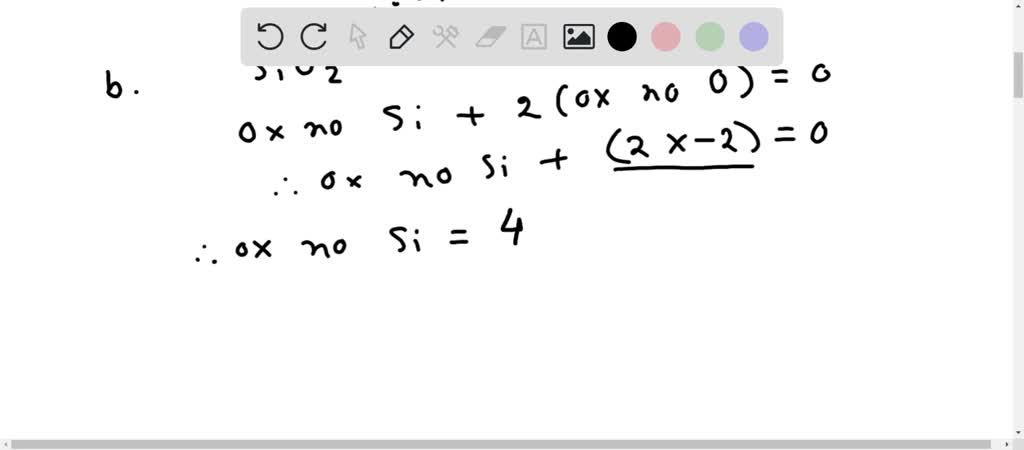
SOLVED: What are the oxidation numbers of: Si in SiO2 N in NH4+, NH3, N2O, NO, NO2-, NO3- Mn in MnOOH C in CH2O, CO2, CH4, C6H6 Ca in Ca2+ Al in

100 Nano-Stories: Silica vs. Silicon + Oxidation of Silica! | by Carlos Manuel Jarquín Sánchez | Medium

Oxidation: any process by which an entity loses electrons. e.g.2Mg o + O 2 o 2Mg 2+ + O 2- H 2 o + F 2 o 2H + F - Mg
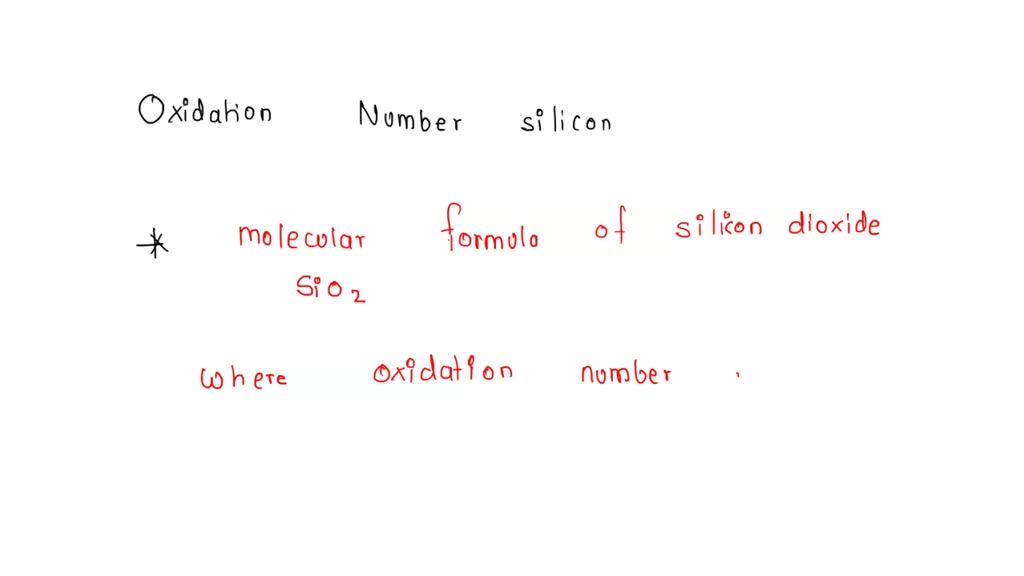
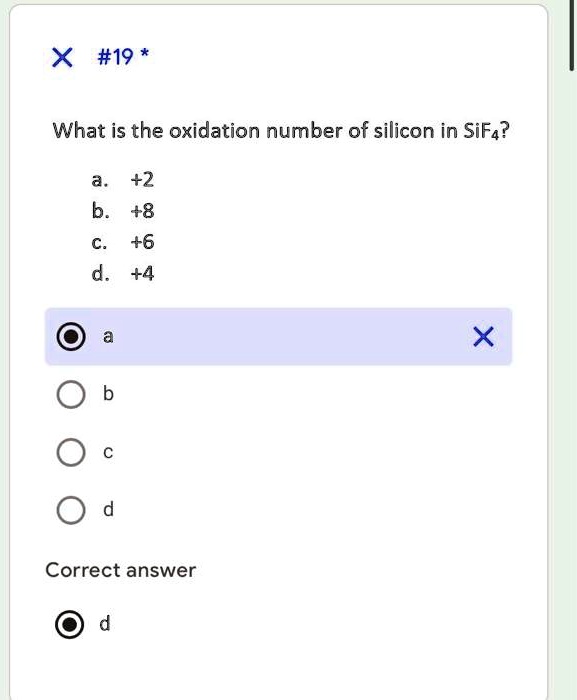
SOLVED: The oxidation number of silicon in silicon dioxide is . Select one: a. +2 b. +4 c. +8 d. +5 e. +6