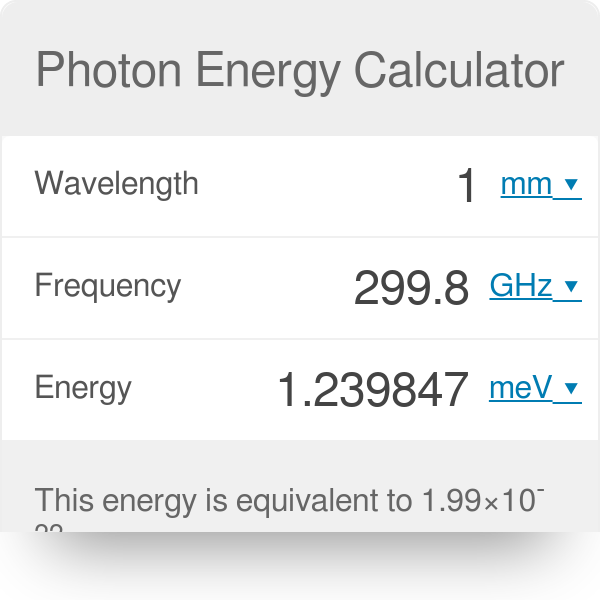
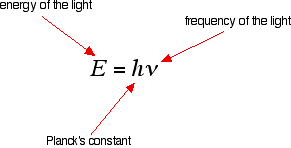
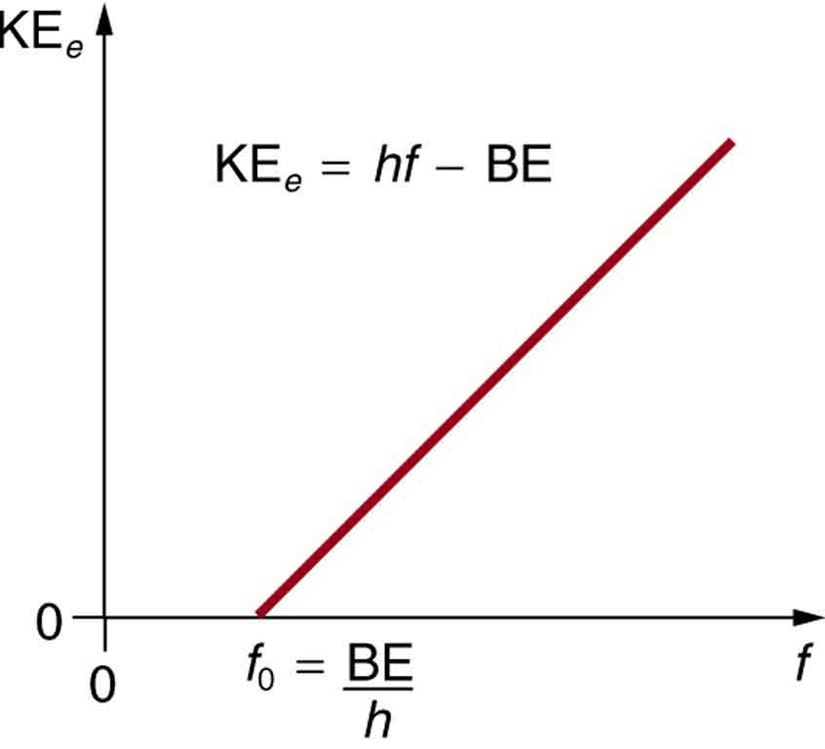
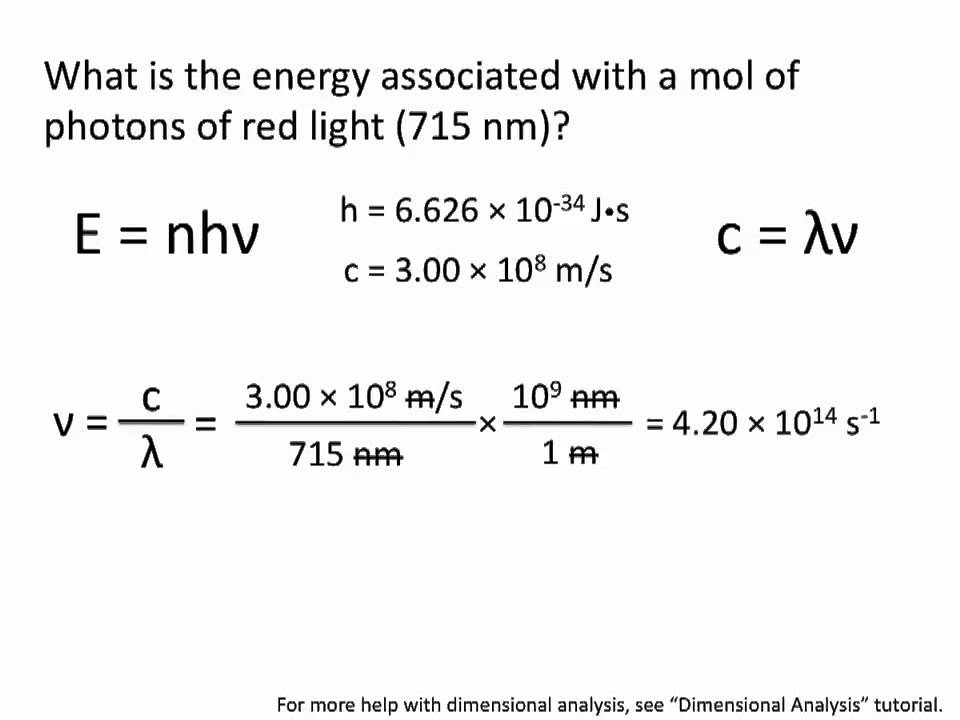
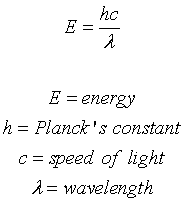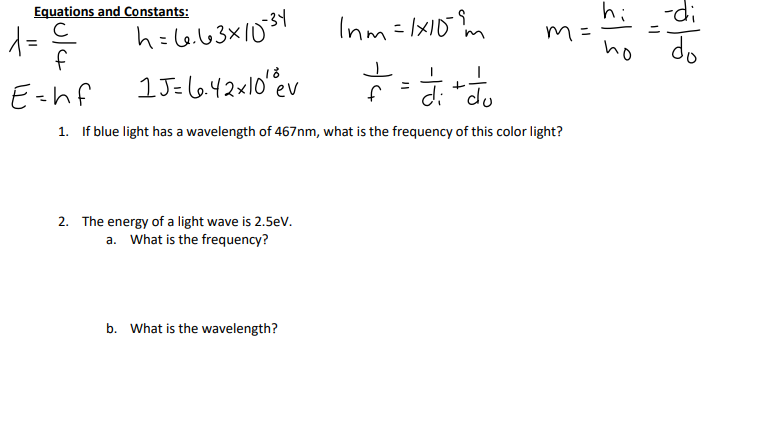SOLVED: Planck's equation relates frequency to photon energy by: E=hv, Where; vis the frequency of the radiation and h is Planck's constant; h = 6.626 x 10-34 J s. If given wavelength;

The relationship between photon energy and wavelength. According to the... | Download Scientific Diagram
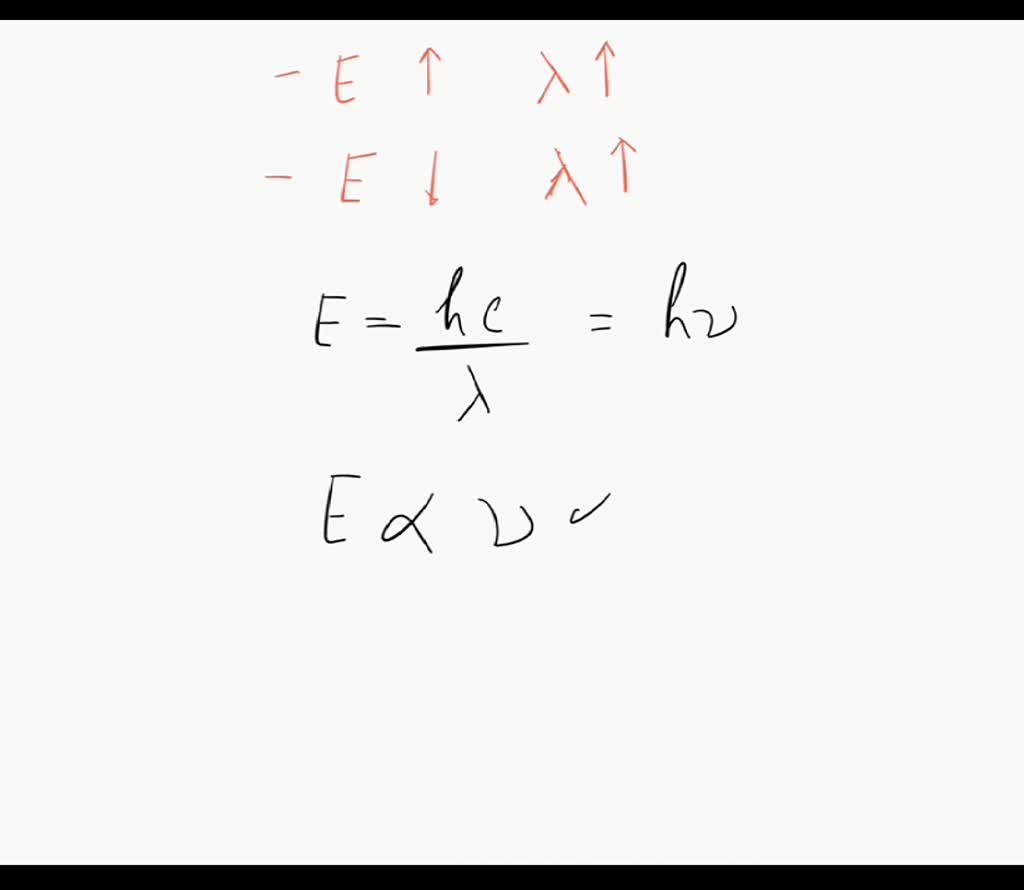
SOLVED: According to the equation for the energy of light, which statement is true? (1) The energy of light increases as its wavelength increases. (2) The energy of light decreases as its